The European REACH regulation (EU-REACH)
Main points
The REACH regulation came into force in 2007. The name is an abbreviation of Registration, Evaluation, Authorization of Chemicals.
The most important provision is that chemicals placed on the market in the EU must be registered by the manufacturer or importer with the European Chemicals Agency (ECHA) with a dossier containing the necessary knowledge and data to assess the safety. The assessment (evaluation) by ECHA may lead to restrictions or even a ban on placing the substance on the market without authorisation. Strict conditions may be attached to such an admission.
The regulation is a European law that is valid in all Member States. Some other countries, such as Norway, also participate. Since Brexit, the UK has its own version: UK-REACH. The law is enforced within the EU by the Member States, which have included provisions for this purpose in national laws.
The following sections discuss the various aspects in more detail.
To which substances does REACH apply?
REACH applies in principle to all chemical substances, but there are exceptions. For example, substances that fall under other laws (e.g. medicines, foodstuffs, waste) are excluded. Polymers are also excluded because the risks are included in the registrations of the monomers of which they are composed. There are also lists of specific exceptions (REACH Annexes IV and V). Substances that are only marketed in very small quantities (less than 1 tonne per year) also do not need to be registered.
To whom does the REACH regulation apply?
The REACH registration duty applies to manufacturers or importers of a chemical substance. So the company that places a substance on the EU market needs to register the substance. In the case of a mixture, the registration obligation applies to all substances in the mixture above a certain threshold. In addition, there are obligations for suppliers, such as distributors. This mainly concerns safety data sheets and risk assessments for the use of a substance that is different from that stated in the REACH registration file.
REACH for importers
The registration duty applies when a substance is introduced into the customs territory of the EU. So when the product is custom cleared. Who is responsible for REACH registration? The REACH Regulation states that the party responsible for the physical introduction into the EU customs territory must hold a REACH registration for the substance(s) in the product. The Incoterms or specific contract clauses might indicate who is responsible for customs clearance, but in practice there are usually several parties involved in the import process. Sometimes it is not clear who exactly has the registration obligation: buyer, seller or one of the intermediate service providers. To avoid problems, it is advisable in such cases to deal with the REACH obligations in the purchase or delivery contract.
There is an exception to the above: if the manufacturer of the product already has arranged a REACH registration, the importers do not need to have their own registration (see below).
REACH for manufacturers
Companies established in the EU that manufacture a substance within the EU will have to register that substance (with some exceptions). The registration obligation also applies to intermediate products, but only if those intermediate products are stored, i.e. not directly converted into another substance.
REACH for manufacturers from outside the EU
The manufacturer of a substance or mixture not established within the EU can appoint a representative who is established in the EU and who acts as the REACH registrant. In that case, importers do not need to have their own registration.
REACH for distributors
If you are a distributor of a substance or mixture, you do not need to have REACH registrations yourself, because that obligation applies to importers and manufacturers. Even if you compose a mixture or formulate a product, there is no registration obligation, because this only applies to the individual substances in the mixture. However, you must ensure that the manufacturers or importers have registered the substances. Your intended use of the product must be included in their registration dossiers. If not, you may need to prepare and submit your own risk assessment to ECHA. In the case of products that contain hazardous substances above certain threshold values, you will also have to have a safety data sheet drawn up.
Collaboration with other registrants
To avoid that every registrant has to conduct all kinds of research themselves, which may also require animal testing, while other manufacturers or importers have already done so, there is an obligation to cooperate. The main reason for this obligation to cooperate is to prevent unnecessary sacrifice of laboratory animals for research. However, other registrants of the same substance are often competitors so how can one cooperate with them? All kinds of rules have been drawn up for this purpose. One of the registrants must act as lead registrant, the others are the co-registrants. The scientific information in the registration dossier is submitted by the lead registrant. The co-registrants can limit their file to company-specific information. However, they must have obtained the right to refer to the information in the lead registrant's dossier. This right must be paid for to cover the costs of scientific research. The REACH Regulation sets rules for distributing such costs among registrants. There are also opt-out options to register without collaboration. If animal experiments are older than a certain period (12 years), the right of the data owners (usually the first registrants) to request money for this lapses. ECHA then provides the right to refer to that data in the registration. ECHA's assistance can also be requested in case of conflicts with other registrants regarding data sharing and costs.
How does that work in practice, this cooperation between competitors? For most substances, REACH consortia have been set up, partnerships of companies that have outsourced the implementation of the obligations to, for example, a consultancy firm. These can be contacted for a quote for the right to use the registration data.
The cooperation is indirectly enforced by ECHA. During submission of the registration file to ECHA, a token is required. This token is generated by ECHA and issued to the lead registrant. This token will only be shared to other registrants if they have made payment for the registration data.
Contents of a REACH registration dossier
The registration dossier usually consists of the following elements.
- identity of the registrant
- chemical identity of the substance
- information on manufacture and use of the substance
- hazard classification and labelling (according to the CLP regulation)
- guidelines for safe use
- physical and chemical properties
- toxicological information (summaries of study results)
- ecotoxicological information (summaries of study results)
- chemical safety report (with exposure scenarios and risk assessments for humans and the environment)
- testing proposals if certain information is not sufficiently known
A complete registration dossier can easily consist of thousands of pages. The information requirements are set out in great detail in the annexes to the REACH regulation and are further explained in ECHA guidance documents. The software package that must be used to prepare registration dossiers (IUCLID) also forces the user to fill in many details.
Fortunately, in most cases a substance has already been registered previously and the new manufacturer or importer can suffice with a more limited dossier.
Assessment of a REACH registration dossier
After submitting the registration, an assessment is carried out by ECHA to determine whether the dossier is complete. The registration number is then assigned. This doesn’t mean that a real substantive assessment has taken place. This is done afterwards on a random basis and may lead to further requests from ECHA and a need to update the file. Sometimes this is even years after the date of registration.
Evaluation of the REACH registrations for a substance
Another type of assessment is whether the risks associated with a substance have been sufficiently investigated and controlled. This is done by an assessment of the content of all registrations for the same substance. ECHA has an action plan for these substance evaluations which is implemented together with national environment and health agencies or institutes. Such evaluations may lead to restrictions or bans (see below).
Restrictions
The restrictions imposed on substances are published in a long list, in Annex XVII of the REACH regulation. A substance may not be placed on the market, manufactured or used unless the conditions stated in the restrictions are met.
Both the European Commission and the Member States can initiate the process to impose a new restriction. The first step is to prepare a file with the data on which the request is based. After an assessment by the ECHA Risk Assessment and Socio-Economic Analysis Committees as to whether the dossier meets the requirements set out in Annex XV of the Regulation, a public consultation will take place. The aforementioned committees then assess the justification and conditions for a restriction, after which the European Commission makes a decision to adapt Annex XVII, which includes all restrictions.
If substances are carcinogenic, mutagenic or toxic to reproduction, and the proposed restrictions are intended for consumers, the European Commission may skip a number of steps in the procedure before amending Annex XVII, allowing such restrictions to be implemented more quickly.
ECHA maintains a list of substances for which a restriction has been requested and this list can be consulted via their website.
Prohibition unless permission (Authorisations)
An even more far-reaching measure is a ban on using or placing a substance on the market, unless an authorisation has been granted. The purpose of the authorization obligation is to prevent substances from being placed on the market if they are considered too dangerous. This goes further than imposing restrictions: it is simply prohibited to place a substance subject to authorization on the market if one does not have an authorization (permission).
The list of substances subject to authorization is formed by Annex XIV of the REACH Regulation. This list may be expanded from time to time by the European Commission, following an approval procedure involving a committee comprising representatives of the Member States, the European Parliament and the Council of Ministers.
ECHA makes proposals for inclusion in Annex XIV based on a list of candidate substances, also called Substances of Very High Concern (SVHC). SVHC substances must be carcinogenic, mutagenic or toxic to reproduction, or persistent, bio-accumulative and toxic (PBT substance) or very persistent and bio-accumulative (vPvB substance), or there must be scientific evidence indicating a high degree of concern. Member States and ECHA may propose to designate such substances as SVHC substances, after which a public consultation will begin.
This step-by-step procedure can be schematized, somewhat simplified, as follows.
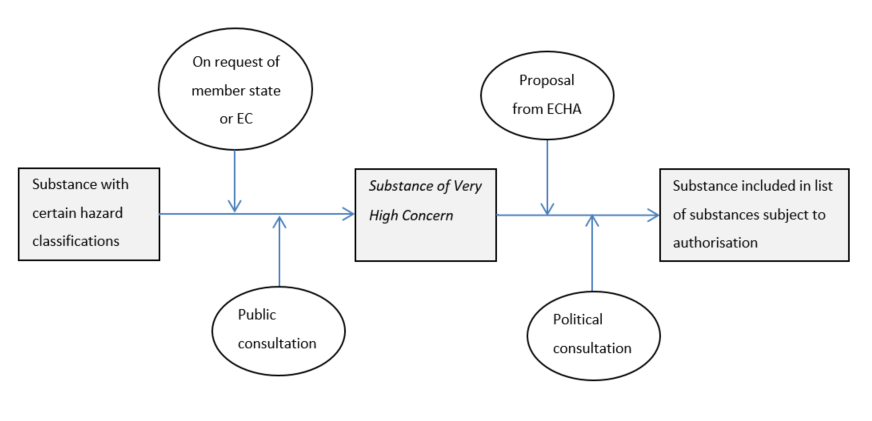
When the decision is made to include a substance in the list of substances subject to authorization, the “sunset date” is determined, i.e. the date by which the substance may no longer be placed on the market without an authorization. Also the date is determined by which authorization requests must be received from manufacturers wishing to continue producing the substance for use after the sunset date. For authorization requests submitted after that date, placing on the market must wait until an authorization has been obtained.
In principle, an authorization will not be granted unless it can be demonstrated that the socio-economic benefits of using the substance outweigh the risks to health and the environment, and there are no suitable alternatives.
An authorization number is assigned to each authorization, which must be stated on the packaging of the substance. Users of a substance subject to authorization must report to ECHA within three months of receiving the substance. This allows compliance with the authorization conditions to be checked.
As mentioned, a substance that is subject to authorization may not be placed on the market without authorization, but there are exceptions. For example, the use for purposes that are already regulated in other legislation, such as crop protection products, biocides, cosmetic products, under certain conditions. Substances that are only used in mixtures below a certain concentration threshold are also exempt. Furthermore, substances used as fuel in engines and combustion plants are exempt from authorization.
Safety Data Sheets
An important obligation for suppliers of chemical products is to provide safety data sheets (also called Material Safety Data Sheets, abbreviated to SDS or MSDS), at least when the product must be classified as hazardous according to the CLP regulation.
Detailed regulations for the contents of the SDS can be found in Annex II of the REACH regulation. The intention is that customers become familiar with the dangers of the product and with the safety measures to manage the risks.
An SDS also shows whether the substances in the product are registered. Then the REACH registration numbers must be stated in section 1 or section 3.
As a distributor of a substance, you have the choice to pass on the SDS from your supplier to your buyers, or to make your own version. Most information can be obtained from the REACH dossier for the substance. But if you put together a mixture, it is more complicated. As a first step you will have to determine the classification according to the CLP regulation and then the other information, such as the hazard properties and the guidelines for safe use. It is work for knowledgeable specialists.
Complementary regulations
The REACH regulation is linked to the CLP regulation, which regulates the classification and labelling of hazardous substances and mixtures and also contains packaging requirements. These two regulations form the backbone of chemical legislation in the EU. The classification, such as “highly flammable” or “possibly carcinogenic”, determines which of the requirements of the REACH regulation do or do not apply to a product.
There are also some complementary regulations, derived from REACH that set additional rules:
Fee regulation This regulation determines the amounts to be paid to ECHA for certain actions such as submitting a REACH registration file.
Implementing regulation on updating registration data. If certain information in the registration dossier is no longer correct, it must be updated without undue delay. This is regulated in art. 22 of the REACH Regulation. To further regulate and specify the period within which changes in the file must be updated, the additional regulation has been introduced. This allows enforcers to better determine whether a company is in violation if registration data no longer matches reality.
The Test Methods Regulation. This describes in detail which international standards may be used for the various tests to obtain the data required for REACH registrations, such as toxicity data.
The implementing regulation for joint submission and exchange of data. Further rules have been drawn up, including how the testing costs must be made transparent and distributed between registrants.
Implementing regulation on the application of provisions after the transition period has expired. This regulation is a legal amendment and stipulates that certain rules still apply despite the fact that a previously set transition period has expired.
Regulation on ECHA's Board of Appeal. The REACH regulation introduced an appeals process and stipulated that there shall be a Board of Appeal. A separate regulation has been introduced for the operation of this Board of Appeal.
References (May 2024)
EU-REACH regulation: EC/1907/2006
https://eur-lex.europa.eu/legal-content/en/TXT/HTML/?uri=CELEX:02006R1907-20231201
CLP regulation: EC/1272/2008
https://eur-lex.europa.eu/legal-content/en/TXT/PDF/?uri=CELEX:02008R1272-20231201
Fee regulation: EC/340/2008
https://eur-lex.europa.eu/legal-content/en/TXT/HTML/?uri=CELEX:02008R0340-20210621
The implementing regulation for joint submission and exchange of data: EU/2016/9
https://eur-lex.europa.eu/legal-content/en/TXT/PDF/?uri=CELEX:32016R0009&qid=1453380621080&from=NL
Implementing regulation on the application of provisions after the transition period has expired: EU/2019/1692
https://eur-lex.europa.eu/legal-content/en/TXT/PDF/?uri=CELEX:32019R1692
Implementing regulation on updating registration data: EU/2020/1435
https://eur-lex.europa.eu/legal-content/en/TXT/PDF/?uri=CELEX:32020R1435
Test Methods Regulation: EC/440/2008
Consolidated version without latest changes: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:02008R0440-20230326
Changes in 2023: https://eur-lex.europa.eu/legal-content/en/TXT/PDF/?uri=CELEX:32023R0464
Regulation on ECHA's Board of Appeal: EC/771/2008
https://eur-lex.europa.eu/legal-content/en/TXT/PDF/?uri=CELEX:02008R0771-20160615
